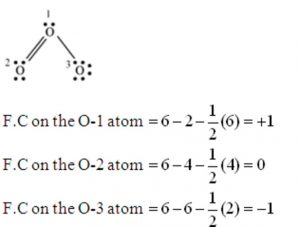 
There are a few rules of thumb that use formal charge that can be used to assist determine which of the various configurations is the most likely for a given molecule or ion: The procedures for constructing Lewis structures may result in more than one conceivable molecular structure in many circumstances – many alternative bonds and lone-pair electron locations, for example, or different groupings of atoms, to name a few examples. The molecular structure of a molecule or an ion refers to the arrangement of atoms within the molecule or ion. Formal charge used to predict the structure of molecules The fact that it is more steady means that it is the most likely. While each of the possibilities results in a formal charge of 0, the first option is the best because it forecasts that there will be no charge in the molecule. Single bonds can be formed between the carbon atom and each oxygen atom (carbon = +2, oxygen = -1 each, formal charge = 0) and between the carbon atom and each nitrogen atom. One or more oxygen atoms may be in a single bond with the carbon atom and another oxygen atom may be in a double bond with the carbon atom (carbon = +1, oxygen-double = 0, oxygen-single = -1, formal charge = zero). For the purpose of determining formal charge, there are three possible ways to draw the Lewis structure for the molecule:ĭouble bonds can be formed between the carbon atom and either or both oxygen atoms (carbon = 0, oxygen = 0, formal charge = zero). Calculation of a Formal Charge in an ExampleĬarbon dioxide, also known as CO2, is a neutral molecule with 16 valence electrons, making it a good example. The following equation is used to compute the formal charge:Īssuming that the atom is isolated from the molecule, eV equals the number of valence electrons in the atom.ĮN is the number of valence electrons on the atom in the molecule that are not bound.ĮB is the number of electrons shared by the bonds that connect the atoms in the molecule to other atoms. For the sake of formal charge, it is assumed that any shared electrons are equally distributed across the two linked atoms. The difference between the number of valence electrons presents in each atom and the number of electrons with which the atom is linked is known as the formal charge of FC. It is possible to compute the formal charge of every individual atom in a molecule using the following equation:įor a neutral atom in isolation (in its ground state), V is the number of valence electrons present L is the number of nonbonding valence electrons present on this atom in the molecule and B is the total number of electrons shared in bonds with other atoms present in the molecule. Whenever a molecule’s Lewis structure (also known as predominant resonance structure) is determined, the structure is chosen so that the formal charge on each of the atoms is as close to zero as possible. The difference between the number of valence electrons present in an atom in its neutral free state and the number of valence electrons present in that atom in its Lewis structure, to put it simply, is the formal charge. 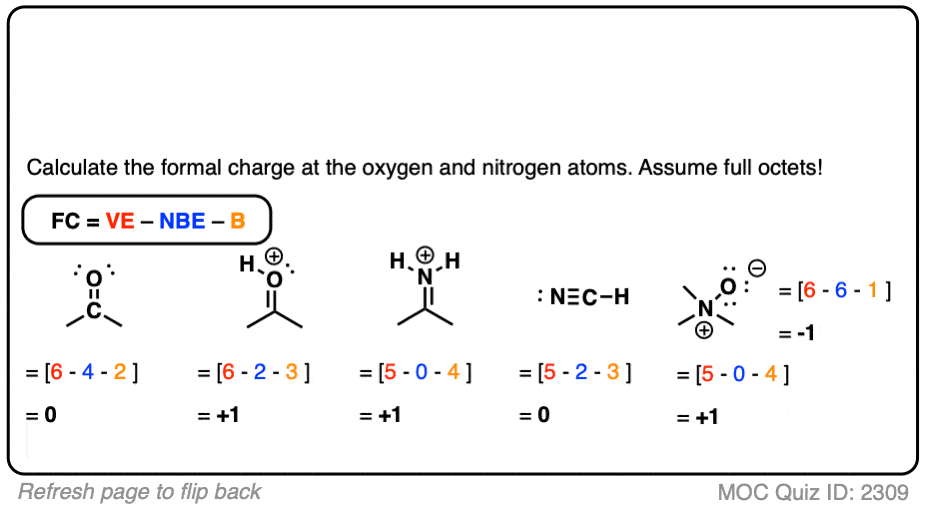
or q) in the covalent view of bonding is the charge assigned to it by the covalent view of bonding, which assumes that electrons in all chemical bonds are shared equally across atoms, regardless of relative electronegativity between them. \) have the same strength and length, and are identical in all other properties.An atom’s formal charge (F.C.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
